If you have ever looked at a battery discharge graph, you may have noticed two different voltage lines a smooth theoretical curve and a real operating curve that dips whenever current flows. At first glance, the difference seems small, but for engineers working with lithium batteries, energy storage systems, or electric mobility, that gap tells an important story. It explains how batteries actually behave in real-world conditions, and why understanding open-circuit voltage (OCV) and cell potential is critical for performance, safety, and long-term reliability.
This blog breaks down these concepts in a practical way not just from a textbook perspective, but from what really matters when designing or operating modern battery systems.
What Is Open-Circuit Voltage (OCV)?
Open-circuit voltage is simply the voltage of a battery when it is at rest — no charging, no discharging, and no external load connected. In this state, the cell reaches electrochemical equilibrium, meaning the voltage reflects the true energy level stored inside.
Think of OCV as the “resting heartbeat” of a battery. It is calm, stable, and unaffected by external stress.
Engineers use OCV primarily for:
Estimating state of charge (SOC)
Cell balancing inside battery packs
Diagnosing ageing or degradation
When a battery sits idle for some time, its measured voltage slowly settles toward the OCV value. That’s why battery management systems often rely on rest periods to recalibrate SOC calculations.
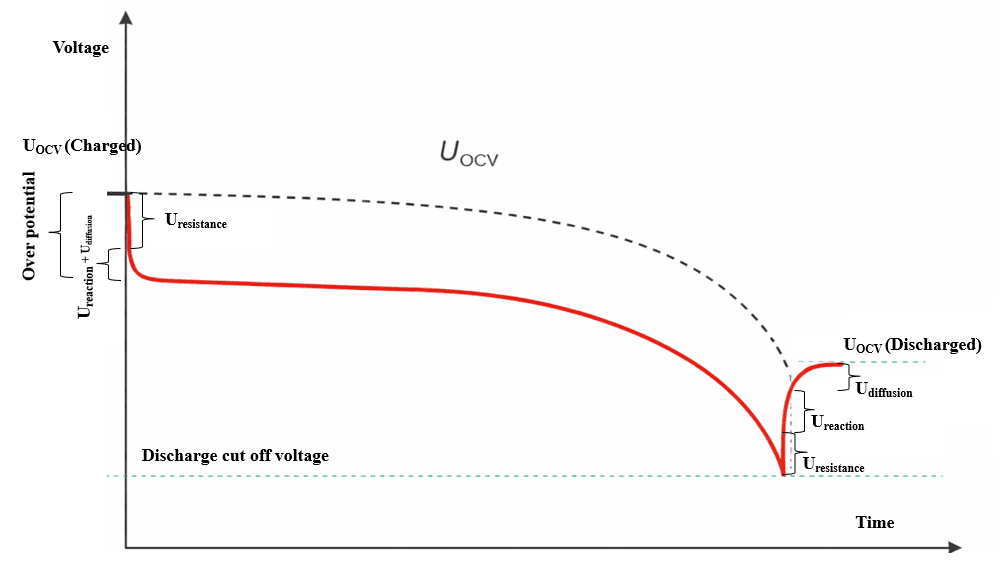
The Real Voltage: Cell Potential Under Load
The moment a battery starts delivering power, its voltage drops below the OCV level. This is completely normal. The drop happens because energy has to travel through internal resistance and electrochemical barriers inside the cell.
This “working voltage” is known as the terminal voltage or cell potential.
Several factors contribute to the difference between OCV and operating voltage:
Internal resistance of electrodes and current collectors
Electrolyte conductivity
Temperature effects
Ageing of the cell
Discharge rate
In simple terms, the harder you push a battery, the more the voltage sags. For example, in high-power applications like BESS, the inverter might demand sudden current bursts. Even though the battery still holds plenty of energy (high OCV), the terminal voltage may dip significantly for a short time.
Understanding the Shape of a Discharge Curve
A typical discharge profile reveals three important regions:
1. Initial Voltage Drop
At the start of discharge, voltage quickly falls from OCV. This is mainly due to ohmic losses, similar to pressure loss when water starts flowing through a pipe.
For engineers, this initial drop helps estimate internal resistance, which becomes a key parameter for system design and thermal management.
2. The Plateau Region
Most lithium batteries, especially LFP chemistry, spend a large portion of their discharge cycle in a relatively flat voltage region. This plateau is beneficial because it delivers stable power output.
However, it also creates a challenge: voltage alone does not accurately indicate how much energy remains. Two different SOC levels can show nearly identical voltages during the plateau, which is why advanced algorithms combine OCV models with current integration (coulomb counting).
3. The End-of-Discharge Knee
Near the end of capacity, voltage drops sharply. This region is critical because operating below the recommended cutoff voltage can permanently damage the cell.
Battery management systems monitor this point carefully and disconnect the load before the voltage falls too low.
Why the Gap Between OCV and Terminal Voltage Is So Important
At first, the difference between OCV and actual voltage might seem like a minor detail, but it directly impacts real system performance.
1. Efficiency and Heat Generation
The voltage loss multiplied by current becomes heat inside the battery. Higher internal resistance means more heat, which reduces efficiency and accelerates ageing.
2. Accurate Energy Prediction
If a system relies only on terminal voltage, it may underestimate or overestimate available energy. Understanding OCV helps create more accurate performance models, especially for large energy storage projects.
3. Safety Margins
The cutoff voltage is based on terminal voltage, not OCV. This means a battery might still hold energy internally even after the system shuts down a fact that engineers must consider during maintenance or diagnostics.
Practical Relevance for Modern Energy Storage Systems
In real industrial applications such as containerized lithium battery systems, rooftop hybrid storage, or grid-support installations the interaction between OCV and cell potential becomes even more significant.
High discharge currents in peak-shaving systems increase voltage sag.
Temperature variations in outdoor installations change internal resistance.
Ageing cells show larger gaps between OCV and operating voltage, reducing usable capacity.
A well-designed battery management system continuously tracks these parameters and adjusts operating limits to protect the cells while maximizing usable energy.
OCV is Theory terminal Voltage is Reality
One of the simplest ways to understand the relationship is this:
- OCV shows what the battery has
- Terminal voltage shows what the battery can deliver right now
Summary
As battery technology moves deeper into renewable energy integration and large-scale storage, understanding voltage behavior is no longer optional it is fundamental. The difference between OCV and actual cell potential explains why two batteries with the same capacity can perform very differently under load.
For engineers, integrators, and even system owners, paying attention to these voltage characteristics helps improve efficiency, extend battery life, and avoid unexpected shutdowns. In the end, a battery is not just a static energy container. It is a dynamic electrochemical system, and the relationship between OCV and terminal voltage is the language it uses to tell us how healthy, efficient, and capable it really is.
